Question 15.5: Draw the structures of the aldehyde (or ketone) and the two ......
Draw the structures of the aldehyde (or ketone) and the two alcohols produced when the following acetals undergo hydrolysis in acidic solution.
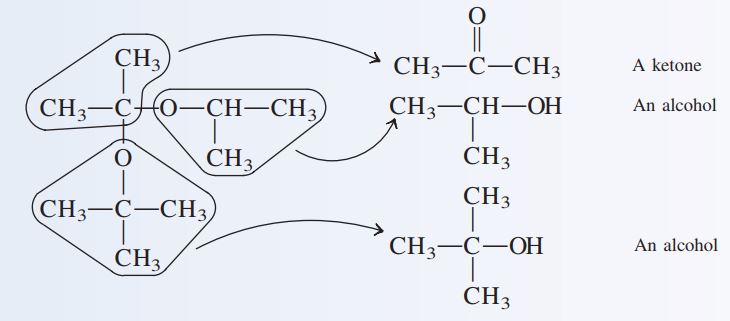
a. \begin{matrix} \quad\quad\quad\quad\quad\quad\quad\quad\quad O-CH_{3} \\CH_{3}-CH_{2}-\overset{|}{\underset{|}{C}}H\\ \quad\quad\quad\quad\quad\quad\quad\quad\quad\quad\quad\quad O-CH_{2}-CH_{3} \end{matrix} b.\begin{matrix} CH_{3} \quad\quad\quad \ \ \\ CH_{3}-\overset{|}{\underset{|}{C}}-O-\underset{|}{C}H-CH_{3}\\ O \quad\quad\quad \ CH_{3} \\ CH_{3}-\overset{|}{\underset{|}{C}}-CH_{3}\quad\quad\quad\quad\quad \\ CH_{3} \quad\quad\quad\quad \end{matrix}
Learn more on how do we answer questions.
a. Each of the alkoxy (—OR) groups present will be converted into an alcohol during the hydrolysis. Because the acetal carbon atom has a H attachment, the remainder of the molecule becomes an aldehyde, with the carbon atom to which the alkoxy groups were attached becoming the carbonyl carbon atom.

b. Again, each of the alkoxy groups present will be converted into an alcohol during the hydrolysis. Because the acetal carbon atoms lacks a H attachment, the remainder of the molecule becomes a ketone.