Effect of Inert Gases on Equilibrium Composition
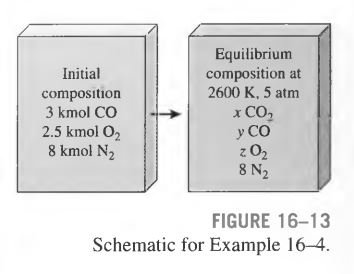
A mixture of 3 kmol of CO, 2.5 kmol of \mathrm{O}_{2}, and 8 kmol of \mathrm{N}_{2} is heated to 2600 K at a pressure of 5 atm. Determine the equilibrium composition of the mixture (Fig. 16-13).
Effect of Inert Gases on Equilibrium Composition
A mixture of 3 kmol of CO, 2.5 kmol of \mathrm{O}_{2}, and 8 kmol of \mathrm{N}_{2} is heated to 2600 K at a pressure of 5 atm. Determine the equilibrium composition of the mixture (Fig. 16-13).