Question 5.SSE.12: A compound D (C8H10O) upon treatment with alkaline solution ......
A compound D ( C_8H_{10}O ) upon treatment with alkaline solution of iodine gives a yellow precipitate. The filtrate on acidification gives a white solid E ( C_7H_6O_2 ). Write the structures of D, E and explain the formation of E.
[IIT, 1996]
Learn more on how do we answer questions.
This reaction indicates the presence of – COCH _3 group in given compound D or presence of – CHOH – CH _3 group in given compound D. On the basis molecular formula of compound D and above reaction,the possible structure is given below:

This compound gives iodoform test as in following reaction. (In it in first step this compound on oxidation with I_2 gives
![]()
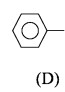 \begin{matrix} CH &-& CH _3&+&3 I _2&+&4 NaOH & \longrightarrow \\ | \\ OH \\\\\\\\ \end{matrix}
\begin{matrix} CH &-& CH _3&+&3 I _2&+&4 NaOH & \longrightarrow \\ | \\ OH \\\\\\\\ \end{matrix}
 \begin{matrix} COONa &+&5 Nai &+&5 H _2 O &+& CHI _3 \downarrow \\ &&&&&& \text {yellowish} \\ &&&&&& \text {crystalline solid}\\ \end{matrix}
\begin{matrix} COONa &+&5 Nai &+&5 H _2 O &+& CHI _3 \downarrow \\ &&&&&& \text {yellowish} \\ &&&&&& \text {crystalline solid}\\ \end{matrix}
